|
A new veterinary medication offers a long-term treatment option for dogs with atopic dermatitis.
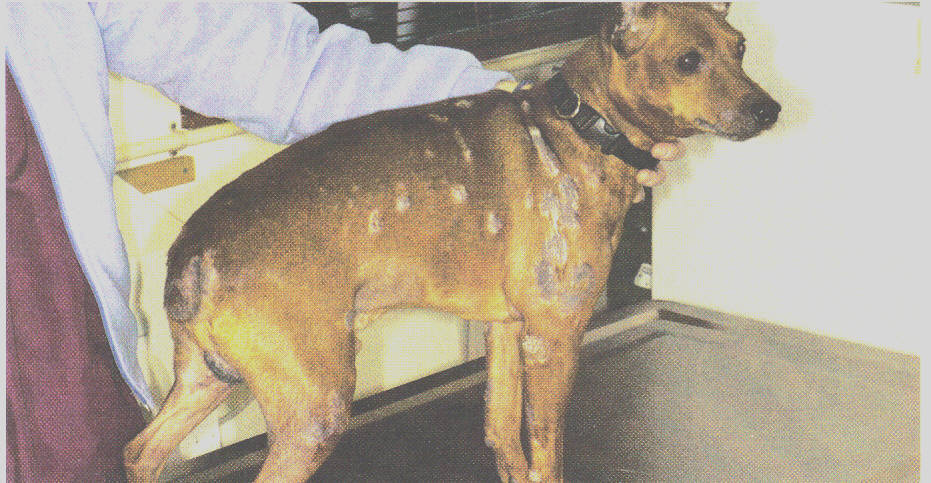
Many dog owners spend considerable time and money treating their dogs' atopic dermatitis, a severe skin condition caused by hypersensitivity to environmental allergens such as pollen, mold and dust mites. In fact, surveys show that dog owners typically spend more than $1,500 on unsuccessful treatments, which traditionally include a mix of corticosteroids, topical medications or antihistamines, according to Novartis Animal Health U.S. Inc. (Greensboro, N.C.).
"Traditional options to control the itchiness and skin lesions associated with canine atopic dermatitis have significant drawbacks," said Helen Power, D.V.M., of Dermatology for Animals (Campbell, Calif.). "Corticosteroids, the most prescribed treatment, are effective in most cases but can cause serious long-term side effects. Antihistamines, another common treatment option, are safe but rarely effective. Many pet owners are using a variety of products and experiencing only marginal satisfaction with the results."
Now, a new veterinary medication developed by Novartis promises safe non-steroidal relief for the 7 million U.S. dogs suffering from atopic dermatitis. Approved by the U.S. Food and Drug Administration, Atopica capsules provide long-term relief without the side effects of corticosteroids. The drug's main ingredient—cyclosporine A—blocks the allergic response that causes discomfort and itching associated with the condition.

"Atopica works differently than the traditional treatments we've been relying on for years," said Power. "Compared to the broad anti-inflammatory action of steroids, Atopica specifically targets the proteins of the immune system that cause the allergic reaction without affecting other organs."
Without treatment, affected pets frequently develop secondary bacterial and yeast skin infections. Their hair may fall out, or their skin may thicken and turn dark with an unpleasant oily residue and odor.
"The ultimate goal is to make each dog as comfortable as possible, but in chronic cases, that hasn't always been an option with existing treatments," Power said. "Now, with allergy advancements like Atopica, dogs and their owners can enjoy control of the allergies without having to trade for the future health of the patient."
ATOPICA is a nonsteroidal oral medication approved by the FDA for the control of atopic dermatitis in dogs. Atopic dermatitis is a lifelong disease that requires longterm treatment. While it can’t be cured, the severe itching and scratching can be treated. Unlike steroids, ATOPICA selectively targets specific immune cells, reducing the occurrence of many side effects. Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Manufacturer:
NOVARTIS ANIMAL HEALTH US, INC. manufactures this medicine for dogs.
What is this medication used for?
Cyclosporine is a strong immunosuppressor. It is useful when hyperactivity of the body’s defense mechanism, the immune system causes more harm than good. This is the case in atopic dermatitis in dogs. Other indications of this drug include dry eye and anal fistulas in dogs. It is also used to prevent rejection of transplanted organs.
Directions:
Atopica capsules should be given on an empty stomach. However, as vomiting is not a rare side effect, sometimes it is necessary to have it given in food. The usual dose is 2-3 mg/lb body weight per day. Some other drugs may slow down the elimination of cyclosporine from the body. In these cases dosage may vary.
Precautions:
Cyclosporine is a strong drug. Liver or kidney dysfunctions, gastric ulcer are conditions when Atopica should be avoided. It should not be given to breeding dogs, pregnant or lactating bitches.
Side Effects:
Vomiting and diarrhea are the most common side effects. Chances of infections increase as a result of the suppressed immune system. Ear and urinary tract infections may occur. Other side effects may include loss of appetite, gastrointestinal problems and gingival hyperplasia.
Storage:
Store in a tight, light proof container. Keep out of reach of children and pets.
|